Carbonation
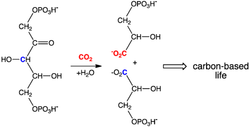
Carbonation is the process of carbon dioxide naturally dissolving into a solution of water under pressure. It is usually found through the creation of soda, tonic water, bottled water, and alcoholic beverages. The chemical formula for carbonation is H2C03 (Carbonic acid) --> CO2 (carbon dioxide) + H2o (water). Effervescence is the escape of gas in a bottled area which contributed to the sound of the fizzing when gas releases from the bottle and an aqueous solution. Carbonated beverages will form bubbles near the top and when the cap is opened initially the carbon dioxide is depressurized to create mass amount of bubbles that will make a fizzing sound. Carbonation forms a certain twist to the taste to give the drink more unique balance and created through the ingredients which makes carbonation. Sprite is the most carbonated made by coke products. Carbonation is measured by temperature and Henry's Law. Henry's Law is at a constant temperature, the amount of a given gas that dissolves and a given type and volume of liquid that is directly proportional to the partial pressure of that gas in equilibrium with that liquid. Henry's Law is shown through coefficients that show changes in density and content within the drink. Carbonated water makes up 20% composition of the soft drink. The bubbles created at the top proves that carbonated water is present and also that there is a chemical reaction transcending in the soft drink. This chemical reaction happens through the formula of carbonation.

